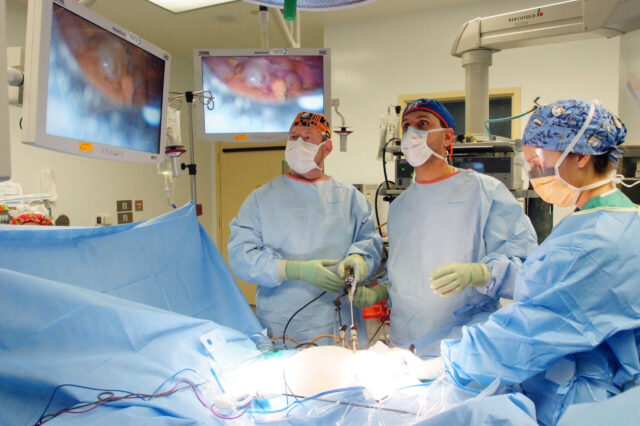
UF Health Surgery in Jacksonville is backed by 30 University of Florida faculty surgeons, 18 physician extenders, 34 resident physicians, and a staff of specialists who combine the brilliance of academic excellence with the advanced clinical skills you would expect from some of the finest physicians in the country.
With our superior team of surgeons and support staff, our success rate is among the highest in the nation. As the region's only academic practice, patients have access to leading-edge treatments, advanced technology and the latest in clinical trials. These resources give patients their best chance to fully restore their health and quality of life.
The UF Health Surgery team encompasses surgical specialists in: